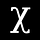The Maxwell-Boltzmann Distribution
An important probability distribution in chemistry and physics
There is an important probability distribution that is well-known in chemistry1 and physics. To the best of my knowledge, it is not widely known outside of these fields.
I am referring to the Maxwell-Boltzmann distribution, which has the following probability density function (PDF):
This distribution is a fundamental concept in statistical mechanics and thermodynamics, describing the distribution of speeds among particles in a gas.
Imagine a container filled with a gas, which consists of many tiny particles (like atoms or molecules) that are constantly moving and colliding with each other. The Maxwell-Boltzmann distribution helps us to understand how the speeds of these particles are distributed at a given temperature.
Not all particles move at the same speed. Some move very slowly, some very quickly, but most have speeds somewhere in between. The Maxwell-Boltzmann distribution provides a mathematical way to describe the probability of finding a particle with a particular speed.
The shape of the distribution depends on the temperature. At higher temperatures, particles have higher average speeds, and the distribution spreads out more.
The Maxwell-Boltzmann distribution is crucial for understanding the behaviour of gases. It predicts how gases will respond to changes in temperature and pressure, and it forms the basis for many other concepts in physics and chemistry, such as the kinetic theory of gases.
To learn more, read the relevant article in Wikipedia.
I now work as a statistician and earned a Master’s degree in statistics. However, I studied chemistry for my Bachelor’s degree.